
25-02-2026
All About Chlor-Alkali Process and Its Applications
Have you ever heard of chlor-alkali? This process has been used in industry since the 19th century. One of the products produced is caustic soda.
So, how does the chlor-alkali process work, and what are its applications in industry? This article explains the ins and outs of the chlor-alkali process. Find out more about it below!
What Is the Chlor-Alkali Process?

Chlor-alkali is an electrolysis process of sodium chloride (NaCl) solution, or table salt, in water that produces three main products: chlorine (Cl2), sodium hydroxide (NaOH) or caustic soda, and hydrogen (H2). This process breaks down the salt solution with electricity to produce these three products.
The process of electrolysis of salt water was first discovered in the early 19th century by William Cruikshank. Later, this process was further developed by many well-known figures, and eventually, a large-scale industrial electrolysis facility was built in England in 1897. This facility produced caustic soda and chlorine.
To this day, the chlor-alkali process is still used in industrial processes. In fact, the technology continues to evolve, with most factories now using the membrane cell process. However, there are also other chlor-alkali technologies, such as mercury cells and diaphragm cells.
Read also: 12 Functions of Caustic Soda for Household and Industry
Chlor-Alkali Technology
There are three main chlor-alkali technologies, namely mercury cells, diaphragm cells, and membrane cells. These three technologies have their own advantages and disadvantages. The following is a detailed explanation:
1. Mercury Cell
The mercury cell is the oldest chlor-alkali technology and is used in almost all parts of the world. However, its use is currently declining due to environmental pollution risks and high energy consumption.
This technology uses a liquid mercury cathode (positive electrode where reduction occurs) for the electrolysis of salt solutions. In this process, the sodium ions formed dissolve into the mercury, forming sodium amalgam (a mixture of sodium and mercury).
The sodium amalgam is then fed into a secondary reactor where it reacts with water. This process produces hydrogen and sodium hydroxide.
The advantage of mercury cell technology is its ability to produce high-purity products. However, this technology uses mercury, which is harmful to the environment.
2. Diaphragm Cell
Diaphragm cells use a polymer or asbestos diaphragm as a separator between the cathode and the anode (negative electrode where oxidation occurs).
The purpose of this separator is to prevent chlorine gas from the anode from mixing directly with hydroxide ions at the cathode. Mixing these two substances can form unwanted hypochlorite ions.
This technology works by allowing the salt solution to flow to the anode, pass through the diaphragm, and move toward the cathode. Then, the water reacts, producing hydroxide ions and hydrogen gas at the cathode. This process forms a liquid sodium hydroxide solution.
The advantage of diaphragm cell technology is its safety compared to mercury cells, as it does not use dangerous chemicals. It is also more environmentally friendly than mercury cells.
However, the sodium hydroxide solution produced remains liquid and contains salt, requiring an additional concentration step. Additionally, the diaphragm, which is in constant use, needs to be replaced periodically.
3. Membrane Cell
The membrane cell is a modern chlor-alkali technology developed in the 1950s. This technology is considered more environmentally friendly because it does not use harmful substances and is more energy efficient.
Membrane cell technology uses a special membrane made of a polymer that allows only positive ions (Na+) to pass. This membrane also serves to retain negative ions (Cl- and OH-). This process produces a purer sodium hydroxide solution because it does not contain salt.
To develop this technology, you need very pure feed water that contains no magnesium or calcium. Furthermore, the membranes used in this technology are very sensitive, which means they require high maintenance costs and advanced care.
Read also: 17 Household Chemical Products and How to Store Them Safely
Chlor-Alkali Process Stages
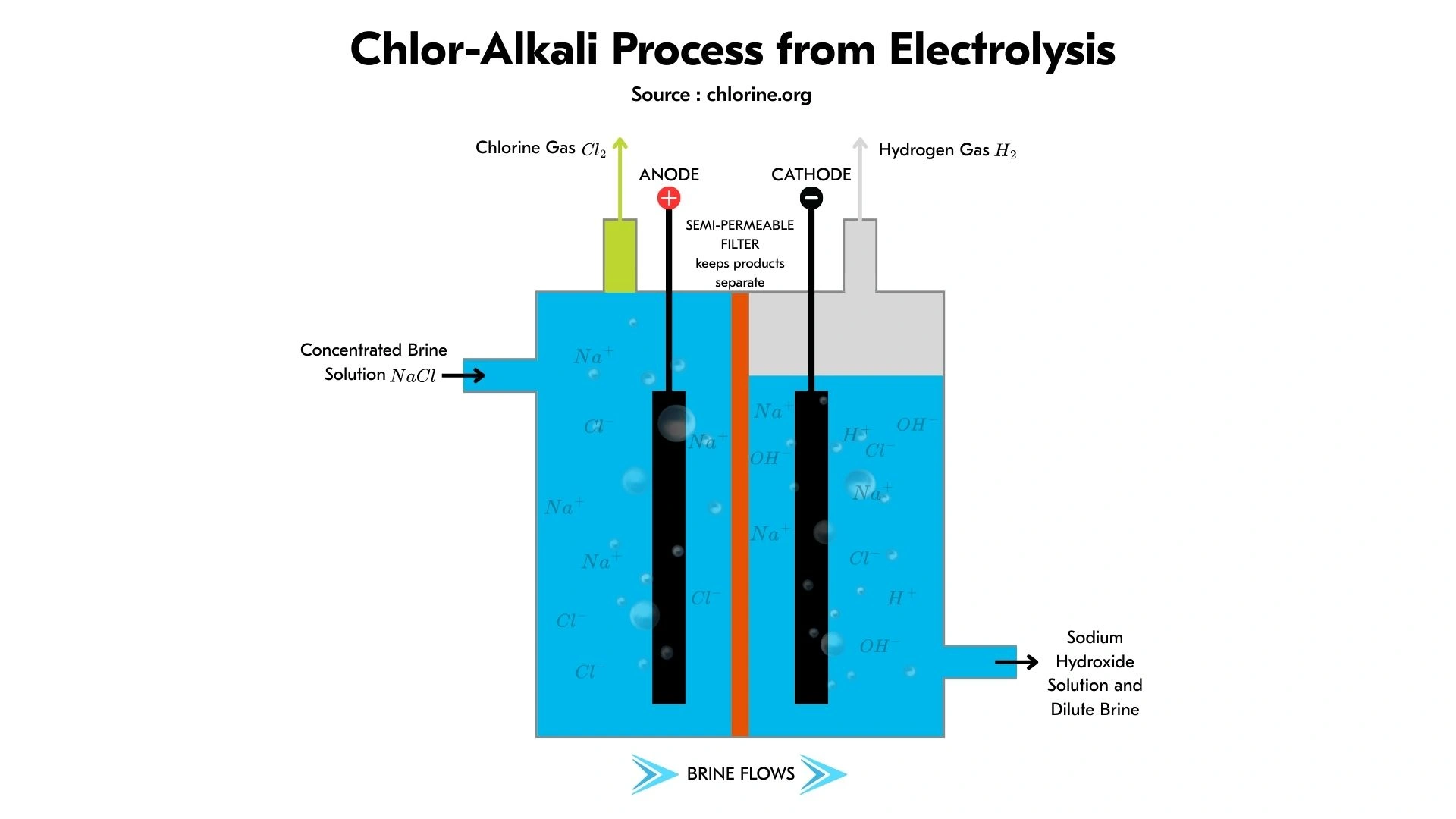
For a better understanding of how chlor-alkali produces chlorine, caustic soda, and hydrogen, here are the stages involved:
- Prepare a pure brine solution. The brine must not contain calcium and magnesium, as these can damage the membrane. To remove impurities from the brine, add sodium hydroxide (to precipitate calcium and magnesium), filter the precipitate, and conduct ion exchange to remove any remaining metal ions.
- Electricity flows through the cell, driving positive ions through the membrane. At this stage, chlorine gas is formed at the anode, while hydrogen and sodium hydroxide are formed at the cathode.
- The chlorine will be collected, while the sodium hydroxide will be purified in accordance with industry specifications.
Applications of Chlor-Alkali

The chlor-alkali process produces three main products: chlorine, caustic soda, and hydrogen. These three products play an important role in various sectors, including the pharmaceutical, water treatment, and chemical industries. The following are the uses of chlor-alkali products:
- Textile industry: Utilizes caustic soda to enhance the strength of fabrics, improve their color absorption, and increase their ability to retain color while preventing color fading. In contrast, chlorine is employed for fabric bleaching.
- Chemical industry: Chlorine and caustic soda are key raw materials. Caustic soda is used to produce soap, detergent, and paper, while chlorine and hydrogen are used to make hydrochloric acid.
- Agricultural industry: Chlorine is found in various pesticides, and caustic soda is used in herbicides.
- Pharmaceutical industry: Chlorine is the key ingredient in the synthesis of medicines, such as drug derivatives and antiseptics.
- Food industry: Chlorine is used to sterilize machines and equipment that come into direct contact with food, thereby preventing contamination. Meanwhile, caustic soda is used to clean vegetables and fruits.
- Solvents: Chlorine is a raw material for industrial solvents, such as chloroform, carbon tetrachloride, and trichloroethylene. These solvents are used to remove oil and synthesize other chemicals.
- Polyvinyl Chloride (PVC): PVC is one of the finished chlorine products that often serves as a raw material for plastic products, such as pipes and cable coatings.
- Disinfectants: Most disinfectant products are made from sodium hypochlorite, which is a form of chlorine. Sodium hypochlorite can also be used as a fabric bleach.
Count on Chandra Asri Group for Your Chlor-Alkali Product Needs!
Primary chlor-alkali products play a vital role in various industries. If your company is one of them, do not worry! Count on Chandra Asri Group, Indonesia's chlor-alkali manufacturer!
As #YourGrowthPartner, we offer chlor-alkali chemical solutions comprising caustic soda, hydrogen, and sodium hypochlorite. Caustic soda is highly needed in downstream industries, such as textiles, paper, and cleaning.
On the other hand, hydrogen is utilized for methanol synthesis, oil refining, and ammonia production. Furthermore, sodium hypochlorite is applied as a disinfectant, particularly in water treatment.
So, secure your company's chlor-alkali product supply with Chandra Asri Group!
Read also: 9 Petroleum Products You Must Know, from Gasoline to Naphtha
.png&w=3840&q=75)
.png&w=3840&q=75)